TRAP/ALP染色试剂盒
- 产品特性
- 相关资料
- Q&A
- 参考文献
TRAP/ALP染色试剂盒
骨组织染色
正常的骨代谢是通过成骨细胞生长与破骨细胞建立骨吸收而维持平衡。成骨细胞的标记酶是碱性磷酸酶(ALP)。破骨细胞的标记酶是抗酒石酸酸性磷酸酶(TRAP)。这两种标记酶在组织切片或者培养细胞过程中,被作为成骨细胞、破骨细胞存在的标记指标。
本试剂盒可通过骨组织切片以及培养细胞中TRAP/ALP酶活性进行组织染色。通过观察成骨细胞和破骨细胞的染色成像,可检查细胞的分化状态和骨组织的分布情况。
◆特点
● 使用时混合3种溶液,可制备TRAP酶活性染色的显色底物溶液。
● ALP酶活性染色使用的是预混物溶液,操作简便。
● TRAP的活性部位呈红紫色,ALP的活性部位呈蓝色(茶褐色),可双重染色。
● 适用于骨组织切片(脱钙GMA树脂包埋切片)以及培养细胞。
◆试剂盒组成
| ● 酒石酸溶液(×10)·······3mL ● 酸性磷酸酶底物液A······30 mL ● 酸性磷酸酶底物液B······3 mL ● 核染色试剂··············10 mL ● 碱性磷酸酶预混物溶液····30 mL
<备注>本产品可对应培养细胞包装24孔板-5次,96孔板-6次。 骨组织载玻片(一个载玻片约使用500μL)包装为60个。 |
 |
◆培养细胞TRAP/ALP酶活性染色案例
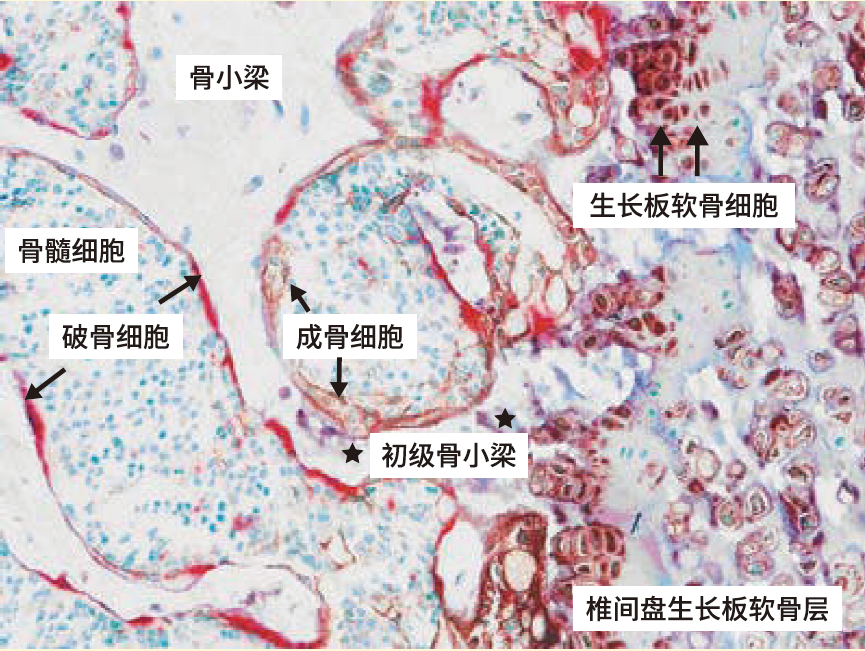 |
● 用TRAP染色剂使破骨细胞呈红色 ● 用ALP染色剂使成骨细胞的细胞膜、软骨细胞和细胞间膜呈茶褐色 ● 用核染色试剂使各种细胞核呈蓝绿色。 |
|
RAW264细胞的TRAP活性染色 |
MC 3T3-E1细胞ALP活性染色 |
◆相关产品
| 产品编号 | 产品名称 | 包装 |
| PMC-AK20-COS | Alkaline Phosphatase Staining Kit 碱性磷酸酶染色试剂盒 |
1 kit |
| PMC-AK04F-COS | TRAP Staining Kit 抗酒石酸酸性磷酸酶染色试剂盒(TRAP) |
10 plate |
详细产品单页请点击下载PDF:TRAP/ALP染色试剂盒
石蜡切片骨相关酶(TRAP,ALP)双重染色
◆实验材料及方法
材料的获取:1年龄小鼠的上、下肢骨以及脊椎骨(切除软组织)
↓
一次固定:4%多聚甲醛溶液·10%福尔马林溶液
二次固定:纯乙醇
部分脊椎骨的非脱钙GMA树脂包埋处理,其他部分做脱钙处理。
↓
脱脂、脱钙:脱钙液是ZnSO4加EDTA溶液。(与其它方法相比)
脱钙装置Histra-DC(普通光)8~16℃连续操作。
↓
脱水、包埋:ETP(Sakura产品)处理16小时,使用融点为58-60℃嵌入式硬石蜡包埋。
部分右上肢和腰椎脱钙GMA树脂处理。
↓
薄切、干燥:制作4μm切片,43℃伸展30分钟后,37℃干燥处理。
↓
染色、封装:TRAP/ALP染色试剂盒(Wako)染色,37℃干燥后,二甲苯透明处理
Malinol封装液封装
↓
镜检,染色性评估
◆标准标本制作
用之前使用的树脂包埋法或冷冻切片法制成的标本作为标准标本,和脱钙石蜡切片相比。即酒精固定小鼠肘关节,用乙二醇甲基丙烯酸树脂包埋,硬组织切片机制作2μm切片,然后进行TRAP和ALP染色(图1)。还有脱钙冷冻切片也可以作为标准标本(图2)。然后,研究或改良固定、脱钙、染色等各阶段双重染色的可行性。
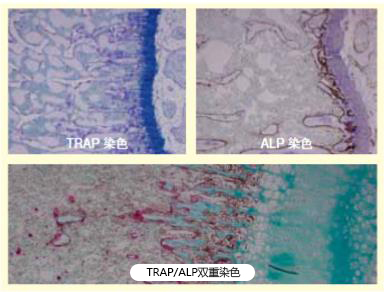
图1:标准标本(GMA树脂包埋)照片是本实验标准标本——小鼠肘关节染色标本。左图为TRAP染色,右图为ALP染色。下图是二者的双重染色。染色效果均佳。以此作为阳性对照,与脱钙石蜡切片一起染色,进行染色性评估。
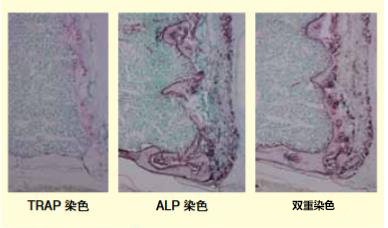
图2:脱钙冷冻切片酶染色—将柠檬酸脱钙的小鼠腰椎和右上肢浸泡在30%蔗糖溶液,后经OCT复合包埋,Tissue‐tekPINO冻结,Cryo制作5μm冷冻切片。再对切片进行酶染色。不仅可进行TRAP染色(左:红色)和ALP染色(中:褐色)的单染色,双重染色(右)也呈阳性。
固定
ALP染色使用不能用福尔马林固定的新鲜材料,如果未在短时间内固定,会导致酶失活,因此推荐80%乙醇固定。①小鼠膝关节不进行一次固定,直接用乙醇直接浸泡进行二次固定组。②小鼠腰椎用福尔马林(4%多聚甲醛溶液)固定16小时后再用乙醇进行二次固定。③小鼠腰椎用福尔马林固定4天后进行二次固定。④临床案例也用福尔马林固定1个月后,再进行二次固定。对上述四种类型进行TRAP·ALP双重染色,研究染色的可行性。如图3所示。本实验证明福尔马林固定从16小时到4天时间范围内均可进行TRAP·ALP双重染色。

图3:福尔马林的1次固定的固定时间和TRAP/ALP的染色性研究—①是未经福尔马林固定,仅用酒精2次固定的样品,ALP染色呈强阳性。TRAP染色呈阴性。②是福尔马林固定16小时和③是固定4天,TRAP/ALP的染色效果均佳。并且,福尔马林固定1个月的临床案例(骨软骨瘤)中TRAP染色呈阳性,但不能ALP染色。
脱钙
ALP酶是含有金属Zn蛋白。通过脱钙处理,酶的组成成分Zn被去除,导致酶失活。为弥补此缺点,需要添加ZnSO4,作为ALP活化剂。换言之,100mL脱钙液需添加0.4mL 1%ZnSO4,来补充Zn离子。并且,作为脱钙剂,在柠檬酸盐酸缓冲液添加Zn的同时,也可尝试添加相同螯合剂EDTA溶液进行研究。另外,也可研究酸性脱钙剂(甲酸福尔马林溶液)能否进行酶反应。结果显示,脱钙液添加EDTA溶液后染色良好。反之,甲酸福尔马林溶液不能进行酶染色。(图4)脱钙方法利用超声波脱钙装置(Histra-DC,正常光)在8~16℃的低温环境下,连续操作3-6天,对样品进行脱钙处理。脱钙后用添加甘氨酸的巴比妥缓冲液(pH7.4)清洗,磷酸钙附着在组织上,防止沉淀。
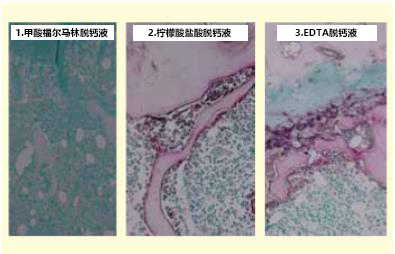
图4:脱钙液的种类不同可否双重染色—左图是选用酸性脱钙溶液中对组织伤害较少的甲酸福尔马林
脱钙溶液脱钙3天(Histra-DC,8~16℃)后的大鼠膝关节。(Histra-DC,8-16℃)TRAP和ALP均不能染色。相对来说,中图、右图均可进行TRAP/ALP双重染色。
染色
染色法是Lorch的Gomori法。本次用的是偶氮染料法和耦合法结合的TRAP/ALP染色试剂盒(Wako,产品编号:294-67001)。对于切片的厚度,Lorch推荐8μm,同时也研究了普通的4μm切片是否能染色、双重染色的顺序应该先染TRAP和ALP中的哪一个、封片剂是否必须选水溶性封片剂等问题。
染色法结果:偶氮染料法中切片过厚导致酶扩散图像。即用TRAP染色骨质有红染倾向,但即使是4μm厚度,只要增强反应条件(反应温度和反应时间),也能充分染色的。换言之,TRAP染色中反应温度从室温调至37℃,反应时间从30分钟调至45分钟或60分钟。ALP染色在37℃反应45分钟至3小时,或者室温下翻译时间至一晚。此结果显示,两者色调平衡,染色效果好。(图5)但是,反应条件的增强导致ALP染色切片产生了很多色素颗粒(图2)。另外,TRAP和ALP的染色顺序哪一个先染色都是可以的。如果先进行ALP染色时,在TRAP阳性部位呈明显的红色,鲜艳处为破骨细胞。获得比较好的标本。但是,ALP的反应产物经过TRAP溶液处理后,会产生白色混浊颗粒,沉淀在组织上。因此,先TRAP染色,接着ALP染色的方法是可行的。封装法是利用甲基绿数秒间核染色处理。水洗后,37℃下干燥,二甲苯浸透,用马里醇(Marinol)永久封存。在浸透前用推荐使用酒精脱水,但是会产生反应产物的溶解、扩散。
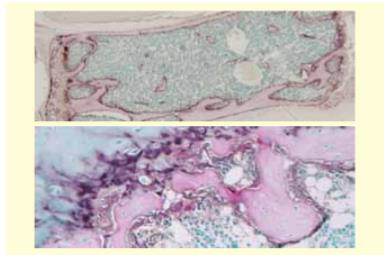
图5:脱钙石蜡包埋切片的TRAP/ALP双重染色—利用1年龄小鼠腰椎的EDTA脱钙石蜡切片,进行TRAP/ALP双重染色。TRAP阳性将破骨细胞染成红色,将强细胞活性的细胞(成骨细胞、软骨细胞)染成褐色。(上:弱扩大,下:强扩大)
◆相关资料

TRAP/ALP染色试剂盒说明书
参考文献
| 1. |
Yamaguchi, S., Aoyama, T., Ito, A., Nagai, M., Iijima, H., Tajino, J., … & Kuroki, H. (2016). Effect of low-intensity pulsed ultrasound after mesenchymal stromal cell injection to treat osteochondral defects: an in vivo study. Ultrasound in medicine & biology, 42(12), 2903-2913. |
| 2. |
Pang, P., Shimo, T., Takada, H., Matsumoto, K., Yoshioka, N., Ibaragi, S., & Sasaki, A. (2015). Expression pattern of sonic hedgehog signaling and calcitonin gene-related peptide in the socket healing process after tooth extraction. Biochemical and biophysical research communications, 467(1), 21-26. |
| 3. |
Hatano, K., Ishida, Y., Yamaguchi, H., Hosomichi, J., Suzuki, J. I., Usumi-Fujita, R., … & Ono, T. (2018). The chemokine receptor type 4 antagonist, AMD3100, interrupts experimental tooth movement in rats. Archives of oral biology, 86, 35-39. |
| 4. |
Azechi, T., Kanehira, D., Kobayashi, T., Sudo, R., Nishimura, A., Sato, F., & Wachi, H. (2013). Trichostatin A, an HDAC class I/II inhibitor, promotes Pi-induced vascular calcification via up-regulation of the expression of alkaline phosphatase. Journal of atherosclerosis and thrombosis, 15826. |
| 5. |
Abe, F., Takahashi, H., & Tanaka, A. (2019). Investigation on the Action and Effect of Culture Supernatant of Human Dental Pulp Stem Cells Using Rats with Medication-Related Osteonecrosis of the Jaw. Journal of Hard Tissue Biology, 28(4), 349-358. |
| 6. |
Kubota, M., Yanagita, M., Mori, K., Hasegawa, S., Yamashita, M., Yamada, S., … & Murakami, S. (2016). The effects of cigarette smoke condensate and nicotine on periodontal tissue in a periodontitis model mouse. PloS one, 11(5), e0155594. |
| 7. |
Maruyama, K., Kawagoe, T., Kondo, T., Akira, S., & Takeuchi, O. (2012). TRAF family member-associated NF-κB activator (TANK) is a negative regulator of osteoclastogenesis and bone formation. Journal of Biological Chemistry, 287(34), 29114-29124. |
| 8. |
Liu, S., Kiyoi, T., Takemasa, E., & Maeyama, K. (2015). Systemic Lentivirus-Mediated Delivery of Short Hairpin RNA Targeting Calcium Release–Activated Calcium Channel 3 as Gene Therapy for Collagen-Induced Arthritis. The Journal of Immunology, 194(1), 76-83. |
| 9. |
Yamasaki, M., Hasegawa, S., Imai, M., Takahashi, N., & Fukui, T. (2016). High-fat diet-induced obesity stimulates ketone body utilization in osteoclasts of the mouse bone. Biochemical and biophysical research communications, 473(2), 654-661. |
| 10. |
Maekawa, S., Katagiri, S., Takeuchi, Y., Komazaki, R., Ohtsu, A., Udagawa, S., & Izumi, Y. (2017). Bone metabolic microarray analysis of ligature‐induced periodontitis in streptozotocin‐induced diabetic mice. Journal of periodontal research, 52(2), 233-245. |
| 11. |
Maruyama, K., Uematsu, S., Kondo, T., Takeuchi, O., Martino, M. M., Kawasaki, T., & Akira, S. (2013). Strawberry notch homologue 2 regulates osteoclast fusion by enhancing the expression of DC-STAMP. Journal of Experimental Medicine, 210(10), 1947-1960. |
| 12. |
Shimomura, S., Inoue, H., Arai, Y., Nakagawa, S., Fujii, Y., Kishida, T., … & Mazda, O. (2018). Treadmill Running Ameliorates Destruction of Articular Cartilage and Subchondral Bone, Not Only Synovitis, in a Rheumatoid Arthritis Rat Model. International journal of molecular sciences, 19(6), 1653. |
| 13. |
Yokota, J., Chosa, N., Sawada, S., Okubo, N., Takahashi, N., Hasegawa, T., … & Ishisaki, A. (2014). PDGF-induced PI3K-mediated signaling enhances the TGF‑β‑induced osteogenic differentiation of human mesenchymal stem cells in a TGF-β-activated MEK-dependent manner. International journal of molecular medicine, 33(3), 534-542. |
| 14. |
Yeom, K. H., Ariyoshi, W., Okinaga, T., Washio, A., Morotomi, T., Kitamura, C., & Nishihara, T. (2016). Platelet‐rich plasma enhances the differentiation of dental pulp progenitor cells into odontoblasts. International endodontic journal, 49(3), 271-278. |
| 15. |
Saita, M., Kaneko, J., Sato, T., Takahashi, S. S., Wada-Takahashi, S., Kawamata, R., … & Nagasaki, Y. (2016). Novel antioxidative nanotherapeutics in a rat periodontitis model: Reactive oxygen species scavenging by redox injectable gel suppresses alveolar bone resorption. Biomaterials, 76, 292-301. |
| 16. |
Wu, Y. H., Taya, Y., Kuraji, R., Ito, H., Soeno, Y., & Numabe, Y. (2019). Dynamic microstructural changes in alveolar bone in ligature‐induced experimental periodontitis. Odontology, 1-11. |
| 17. |
Li, P., Honda, Y., Arima, Y., Yasui, K., Inami, K., Nishiura, A., … & Matsumoto, N. (2016). Interferon-γ enhances the efficacy of autogenous bone grafts by inhibiting postoperative bone resorption in rat calvarial defects. Journal of prosthodontic research, 60(3), 167-176. |
| 18. |
Li, P., Hashimoto, Y., Honda, Y., Arima, Y., & Matsumoto, N. (2015). The effect of interferon-γ and zoledronate treatment on alpha-tricalcium phosphate/collagen sponge-mediated bone-tissue engineering. International journal of molecular sciences, 16(10), 25678-25690. |
| 19. |
Tezuka, R., & Tanaka, A. (2016). Jawbone Changes in Sodium Zoledronic Acid-and Dexamethasone-Treated Rats. Journal of Hard Tissue Biology, 25(4), 383-394. |
| 20. |
Yamada, K., Tsuji, T., & Kunieda, T. (2013). Phenotypic Characterization of Ggt1dwg/dwg Mice, a Mouse Model for Hereditary γ-GlutamylTransferase Deficiency. Experimental animals, 62(2), 151-157. |
| 21. |
Fukushima, H., Shimizu, K., Watahiki, A., Hoshikawa, S., Kosho, T., Oba, D., … & Okabe, K. (2017). NOTCH2 Hajdu-Cheney mutations escape SCFFBW7-dependent proteolysis to promote osteoporosis. Molecular cell, 68(4), 645-658. |
| 22. |
Kawada, S., Wada, E., Matsuda, R., & Ishii, N. (2013). Hyperbaric hyperoxia accelerates fracture healing in mice. PloS one, 8(8), e72603. |
| 23. |
Ikawa, H., Moroi, A., Yoshizawa, K., Saida, Y., Hotta, A., Tsutsui, T., … & Saito, Y. (2017). Bone regeneration enhancement by ultra-violet (UV) treatment for uHA/PLLA absorbable mesh. Journal of Cranio-Maxillofacial Surgery, 45(5), 634-641. |
| 24. |
Sato, M., Asada, N., Kawano, Y., Wakahashi, K., Minagawa, K., Kawano, H., … & Katayama, Y. (2013). Osteocytes regulate primary lymphoid organs and fat metabolism. Cell metabolism, 18(5), 749-758. |
| 25. |
Maruyama, K., Fukasaka, M., Uematsu, S., Takeuchi, O., Kondo, T., Saitoh, T., … & Akira, S. (2015). 5-Azacytidine-induced protein 2 (AZI2) regulates bone mass by fine-tuning osteoclast survival. Journal of Biological Chemistry, 290(15), 9377-9386. |
| 26. |
Okuda, T., Naruo, M., Iijima, O., Igarashi, T., Katsuyama, M., Maruyama, M., … & Haseba, T. (2018). The Contribution of Alcohol Dehydrogenase 3 to the Development of Alcoholic Osteoporosis in Mice. Journal of Nippon Medical School, 85(6), 322-329. |
| 27. |
Gunji, H., Kunimatsu, R., Tsuka, Y., Yoshimi, Y., Sumi, K., Awada, T., … & Yanoshita, M. (2018). Effect of high‐frequency near‐infrared diode laser irradiation on periodontal tissues during experimental tooth movement in rats. Lasers in surgery and medicine, 50(7), 772-780. |
| 28. |
Takano, A., Fukuda, T., Shinjo, T., Iwashita, M., Matsuzaki, E., Yamamichi, K., … & Nishimura, F. (2017). Angiopoietin-like protein 2 is a positive regulator of osteoblast differentiation. Metabolism, 69, 157-170. |
| 29. |
Tsuchiya, T., Sakai, A., Menuki, K., Mori, T., Takeuchi, Y., Kanoh, S., … & Nakamura, T. (2013). Disruption of aldehyde dehydrogenase 2 gene results in altered cortical bone structure and increased cortical bone mineral density in the femoral diaphysis of mice. Bone, 53(2), 358-368. |
| 30. |
Ishikawa, J., Takahashi, N., Matsumoto, T., Yoshioka, Y., Yamamoto, N., Nishikawa, M., … & Yamamoto, A. (2016). Factors secreted from dental pulp stem cells show multifaceted benefits for treating experimental rheumatoid arthritis. Bone, 83, 210-219. |
| 31. |
Suzuki, K., Anada, T., Miyazaki, T., Miyatake, N., Honda, Y., Kishimoto, K. N., … & Suzuki, O. (2014). Effect of addition of hyaluronic acids on the osteoconductivity and biodegradability of synthetic octacalcium phosphate. Acta biomaterialia, 10(1), 531-543. |
| 32. |
Tamura, Y., Kawao, N., Yano, M., Okada, K., Okumoto, K., Chiba, Y., … & Kaji, H. (2015). Role of plasminogen activator inhibitor-1 in glucocorticoid-induced diabetes and osteopenia in mice. Diabetes, 64(6), 2194-2206. |
| 产品编号 | 产品名称 | 产品规格 | 产品等级 | 产品价格 |
| 294-67001 | TRAP/ALP Stain Kit TRAP/ALP双重染色试剂盒 |
60次 | 病理研究用 | - |